P4SPR – Onsite testing of RDX in environmental waters
Onsite testing of RDX in environmental waters (Small molecule detection)
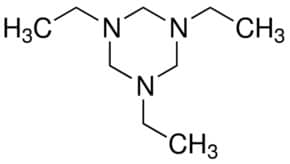
Energetic materials (commonly known as explosives) such as 1,3,5-trinitroperhydro-1,3,5-triazine (RDX)(Figure 1) has been used extensively in the manufacturing of munitions, and accounts for a large part of the explosives contamination at active and former military installations in various parts of the world. Most RDX are not significantly retained by soils, and biodegrade very slowly. As a result, RDX can easily percolate through the ground to contaminate ground water which serves as drinking water for surrounding populations. RDX is not only classified as potentially carcinogenic but it can also damage the nervous system if inhaled or ingested. Hence, continuous monitoring of RDX levels in ground water would be ideal for public safety to reduce RDX exposure to the population and limit its potentially adverse health effects.
Introduction
Currently, the EPA method 8330b specific for detection of energetic materials in environmental samples is based on HPLC-UV in centralized labs. Although sensitive, this method is time intensive as it requires sampling, transportation, storage and sample preparation prior testing, Even though the lead time for the process requires at least 24 hours, days or weeks between sampling and testing are not unusual. Field testing of RDX using label-free sensing methods could potentially cut the sampling to result time from days to hours and significantly reducing the risk of RDX exposure to the population in areas of concern.
Surface Plasmon Resonance (SPR) has proven its label-free sensing and real-time monitoring benefits in biosensing. New applications of SPR, especially in portable format, are being developed to expand these benefits to environmental monitoring of harmful contaminants.
In this application note, we will report a combination of innovative portable SPR device and highly specific RDX recognition deployed on the field to monitor RDX in ground water near downgradient wells. Different analytical parameters were characterized including sensitivity, selectivity, limit of detection and influence of temperature proving the potential suitability of portable SPR as an alternative for testing faster than HPLC-UV methods in centralized labs.
Experimental Setup
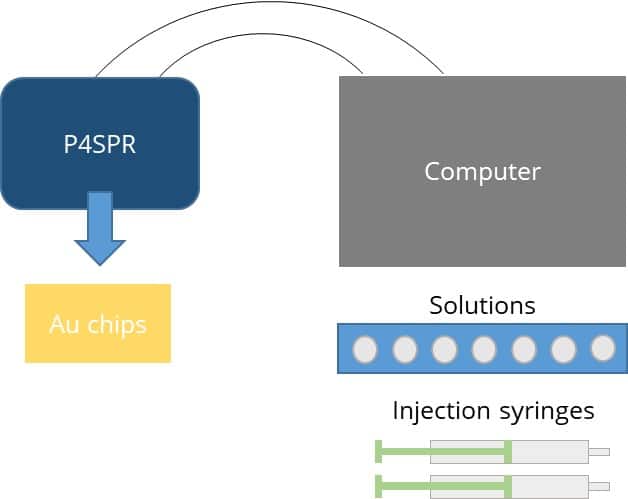

Figure 2 – (left) Schematic of P4SPR set up and right) actual field set up in the back of a pickup truck.
The P4SPR was deployed on sampling sites during both Canadian winter and summer time (-20ᵒC and 30ᵒC). The temporary field set up for the device was on a table or a mat (Figure 2) to shelter it from snow or rain. The device was powered by a laptop via USB. The laptop battery was backed-up by a generator. Well water samples were drawn in two stage using peristaltic pumps to control the flow at the device. First well water samples was pumped into a large collection bucket. Then, water from the collection bucket was pumped into the P4SPR. In the P4SPR, water samples flowed into the 3-channel part of the microfluidic onto the RDX-selective SPR sensor while an uncontaminated water sample was used as reference in the fourth channel to correct for temperature variations (Figure 3). Therefore, each sample was measured in triplicate and corrected in real time.
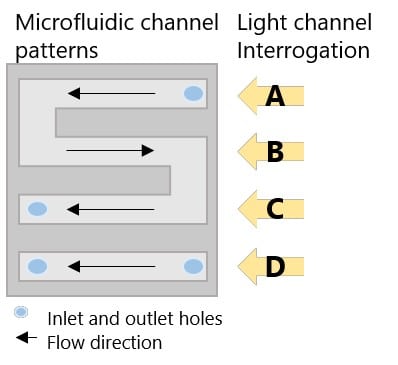
Figure 3. Schematic of 4-channel microfluidic cell and interrogation channels.
Experimental procedures
The sensor chips were functionalized with a Molecular Imprinted Polymer (MIP) selective for RDX by crosslinking a bisaniline polymer with Au nanoparticles (Figure 4). The SPR sensors were first validated in the laboratory with aqueous solutions of RDX. The effect of temperature on the SPR sensitivity was measured with a calibration curve from 1 nM to 50 nM as an environmentally relevant range was then measured in the laboratory from 2°C to 36 °C. For field analysis, the standards were prepared in uncontaminated water upstream.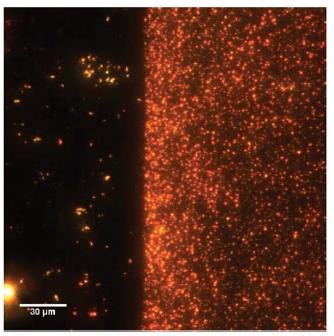
Figure 4 – (left) Dark-field image of no MIP (right) MIP on an ITO slide. The orange dots are gold nanoparticles.
The contaminated samples were analyzed as collected from the well in reference to purified water signal. Furthermore, the response from each sensor was normalized with the response of a 10 nM RDX standard.
Results and Discussion
Analytical validation in the lab Prior field testing, the SPR method was first validated in the laboratory. The sensitivity of the SPR sensor was measured with RDX solutions of 1 pM to 10 nM flowed successively on the sensor at 1 mL/min (Figure 5, top).
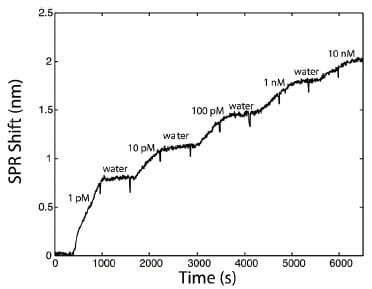
Figure 5a SPR sensorgram for the calibration of the sensor for RDX detection.
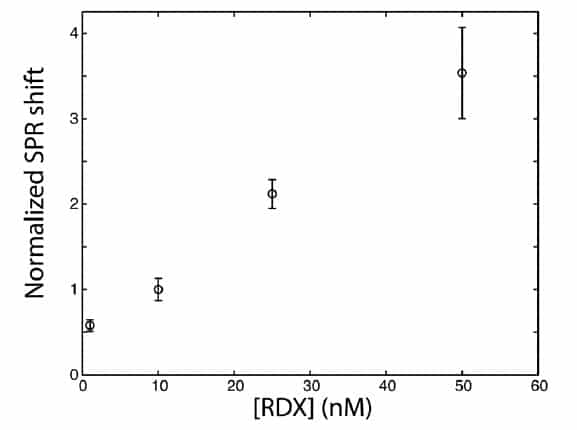
The SPR response was normalized against a 10 nM standard. The error bars represent standard deviation on a triplicate measurement (n=3).
Given field trips would happen at opposite seasons over a wide temperature range, the method was developed to account for and correct for temperature effects on the SPR response in laboratory conditions. The main temperature effect was observed on the SPR sensor sensitivity. However, normalizing calibration curves with the SPR signal at 10 nM for each curve provided a more consistent sensitivity for concentrations above 0.1 nM within the relevant temperature range (Figure 6). This normalising method was applied in the field in subsequent tests.
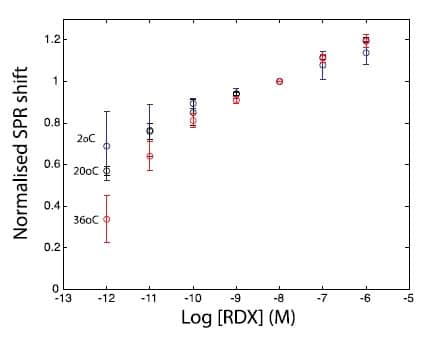
Figure 6 – Normalised calibration curves for RDX at different temperatures against SPR response at 10 nM.
Field SPR testing of RDX
The current field testing approach with SPR requires minimal infrastructure. A temporary shelters such as a tent, trailer or the tailgate of an SUV have proven sufficient to deploy the P4SPR on sampling sites (Figure 7). It took up to 90 minutes from on-site arrival to complete SPR analysis at each well. This includes the setup of the sampling system and P4SPR, equilibration with distilled water and uncontaminated water, measurement of the sample as well as recalibration. In comparison to the EPA standard test, the HPLC-UV method, the SPR field test saved time on sample transportation and preparation. The field SPR method is significantly faster and more suitable for frequent monitoring of environmental samples especially in remote areas.

Figure 7 – Photographs of P4SPR deployed in different season.
In addition, data generated with the field SPR methods showed an excellent correlation with the HPLC EPA method 8330b. This proved the high potential of the field SPR method to not only serve as on-site screening tool for the centralized test but also a potential alternative to it.
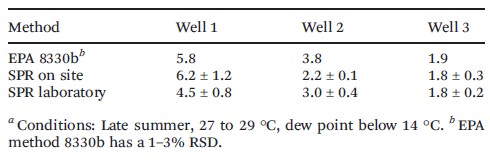
Table 1 – Comparative study of the SPR method and the EPA method 8330b with different wells sampled on site. Concentrations reported are in ppb. 2 ppb corresponds to about 10 nM.
The P4SPR advantage
Affinité instruments’ P4SPR is light-weight, compact and portable, suitable for onsite testing in a range of environmental conditions. The minimal requirements for setup space and signal stabilization in the field dramatically reduce the sampling to result time against conventional methods such as HPLC-UV. The selectivity of SPR sensors can be tuned to capture an analyte of interest using different surface chemistries. Additionally, SPR sensors are capable of direct detection, thus requiring no sample preparation, which make them a versatile sensor.
Conclusion
This study has demonstrated that the P4SPR has offered exceptional analytical performance for the analysis of RDX onsite at the sampling wells. With excellent correlations to the standard HPLC method as well as P4SPR analysis of the samples in the laboratory, users could confidently bring P4SPR onsite for rapid testing of energetic materials in remote areas. The portability and robustness of the instrument have proven it to be a feasible option for environmental monitoring with opportunities of greatly expanding its applications for other contaminants.
About Affinité Instruments
Established in 2015 as a spin-off of the Université de Montréal, Affinité instruments’ foundation is built deep knowledge accrued on over a decade of research in SPR. The commercialization of promising innovations is spearheaded by a diverse leadership experienced in business, science and engineering.
