| Adsorptive | Boiling point T / K | ρLiquid / g cm-3 | Critical diameter of atom/molecule / nm /3/ | am for BET surface from ρLiquid *4 / nm2 | p0 / kPa | Non-ideality factor Fni of the adsorptive *3 |
Ar | 87 | 1.39 | 0.34 | 0.143 | 101 | 4.27·10-5 |
Kr | 120 | 2.41 | 0.36 | 0.163 | 101 | 3.83·10-5 |
Xe | 165 | 2.94 | 0.41 | 0.192 | 101 | 3.71·10-5 |
SF6 | 209 | 1.91 | 0.67 | 0.276 | 101 | 3.87·10-5 |
CH4 | 112 | 0.42 | 0.40 | 0.174 | 101 | 4.26·10-5 |
Ethane | 184 | 0.54 | 0.44 | 0.223 | 101 | 3.86·10-5 |
Propane | 231 | 0.58 | 0.49 | 0.275 | 101 | 3.93·10-5 |
n-Butane | 273 | 0.60 | 0.49 | 0.322 | 101 | 4.05·10-5 |
| O2 | 90 | 1.14 | 0.28 | 0.141 | 101 | 4.09·10-5 |
N2 | 77 | 0.81 | 0.30 | 0.162 | 101 | 5.21·10-5 |
| CO2 | 195*1 | 1.26 | 0.28 | 0.164*2 | 188*2 | 2.09·10-5 |
| Ethene | 196 | 0.58 | 0.39 | 0.203 | 101 | 3.71·10-5 |
| CO | 82 | 0.94 | 0.37 | 0.147 | 101 | 5.11·10-5 |
| NH3 | 240 | 0.70 | 0.29 | 0.128 | 101 | 4.17·10-5 |
| iso-Butane | 261 | 0.59 | 0.51 | 0.325 | 101 | 1.71·10-5 |
3P Instruments
cryoTune series
Gas Sorption Analyser
- Temperature Range 77 K – 323 K
- Easy-to-use software
- Up to 3 devices can be controlled at once

3P cryoTune offers gas sorption measurements
Realisation of measuring temperatures for sorption experiments from 77 K – 323 K
The cryoTune series opens new horizons for gas sorption measurements. It offers the possibility for almost all volumetric sorption instruments, no matter if newer or older production dates, to follow the ISO 9277 /1/ for BET measurements as well as the IUPAC recommendation for micropore characterization /2/. Both publications explain that noble gases, unlike the nitrogen molecule, have a spherical symmetry and no quadrupole moment. Therefore, the adsorption of noble gas atoms results from a pure van-der-Waals interaction and the atoms do not have different orientations on the adsorbent surface.
-
Technical Specs
- Temperature range 77 K – 323 K
- Temperature stability ± 0,004 K (standard deviation)
- Easy-to-use software for data acquisition, display, analysis and archiving
- Up to three cryoTune devices can be controlled by the software via a single USB port
- The universal cryoTune can be used with any commercial gas sorption device that accepts external temperature control for sorption measurements
-
Absorptives
*1…sublimation point
*2…p0 and ρLiquid of the undercooled liquid
*3…mean value from van der Waals- und Bethelot equations
*4…calculated according to:
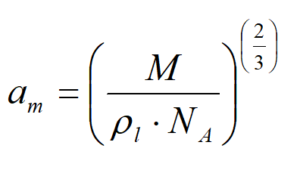
Applications
Techniques
Models
Although a cryoTune can work as stand-alone without a PC, it is delivered with software that can display and record the real time temperature. The cryoTune series consists of three versions, which can be purchased individually or as a package. All models are connected to a measuring station of your sorption device. If you have a multi-station device in use, it is possible to connect several cryoTunes.
The cryoTune 77 is an option for sorption experiments at 77.35 – 78 K with a temperature control accuracy of better than 0.004 K for realizing temperature-stable, isothermal measurements with nitrogen and other gases. Particularly useful for krypton, argon or methane when saturation vapor pressure cannot be measured during measurement and accordingly cannot be corrected. The cryoTune 77 significantly increases measurement accuracy and reproducibility of measurements close to 77 K, due to the exact temperature control no p0 correction is necessary.
The cryoTune 87 covers the temperature range 82 – 135 K, making the cryoTune 87 the standard option for Ar 87 K measurements for surface and pore analysis recommended in ISO 9277 and IUPAC recommendation of 2015.
The cryoTune 195 is the cryoTune version for isotherm measurements in the range of 180 – 323 K. This allows CO2 measurements in particular, but also measurements of other adsorptives such as ethane or propane at their boiling temperatures. For such measurements you neither need a refrigeration thermostat, nor a dry ice refrigeration mixture nor a double-walled additional Dewar. Among others, the cryoTune 195 enables the following CO2 measurement possibilities:
- CO2 adsorption at 273 K
- CO2 adsorption at 195 K up to a relative pressure of ca. p/p0= 0.5 (the entire micropore filling including the BET range can be detected, e.g. for ultra-micropores, where N2 at 77 K is not applicable)
- Measurement of isotherm sets at different temperatures for thermodynamic evaluations, e.g. in the ranges around 273 K or 195 K or over the entire temperature range
The cryoTune 120 completes the cryoTune series and enables long-term measurements especially in the range 115 K (boiling temperature of krypton) over 165 K (boiling temperature of xenon) up to 230 K. The combination cryoTune 87 and 120 thus allows the use of three noble gases at their boiling temperature: Argon 87 K, Krypton 120 K and Xenon 165 K, as well as a multitude of other measurement possibilities with Ar, O2, methane, ethane or propane.
Not sure if it’s the right instrument?
No worries, send us a sample and we will test it for you
Send a sample
-
Outstanding service

I have been using Meritics for a number of years and they always provide outstanding service and support.
They have a rapid response turnarounds and are very knowledgeable of the techniques and products they provide.LeanneB-375
-
Good service, very helpful

We use Meritics for the PM service and installation of all our FlowCams . Megan has been coming to our site for a number of years. She is very efficient in her work, but most of all Megan is very helpful. We have had issues with one of instruments after the install and Megan did her utmost to try and solve it while on site, even though it wasn’t solved Megan has been in contact with the supplier of the instrument to try and solve this problem for us.
SophieH-2201
-
Job done in a friendly manner

Booking engineer visits is always easy. David Lowe, Meritics engineer is always very friendly.
ThierryN
-
Service/calibration visit

Excellent service, arrived on time, polite, friendly and informative.
NathamM-270
-
5 Stars

Excellent service from an excellent company. Would recommend them to anyone.
BarryH-480
-
Good Service

David, our service engineer as usual was good at communicating and setting up the recent service appointment, he arrived on time, did the service promptly and informed me of any problems. Friendly and helpful
TomA-446
-
Superb and prompt service

I have used this company through the laboratory I work for frequently. We specialise in soil analysis, and Meritics were chosen as they have extensive knowledge of the instruments we use. We have several laser particle diffraction analysers with a service contract, and service is always very prompt and meticulous….
JamesG-2483
-
Good experience over two years

Meritics have covered the annual servicing of our Multisizer 4e instrument since we purchased it from them just over two years ago. The application scientist who performed the servicing was friendly, knowledgeable, and extremely helpful, happily answering a barrage of questions about how best to use and maintain the instrument. All my interactions with Meritics staff have been positive. I would happily recommend them.
RajG-62
-
Fully committed to customer service

Service second to none, both units serviced and all questions answers by engineer while on site.
RichardA-786
-
Highly recommend Meritics for equipment purchases and aftercare services

After owning one of our instruments for over 8 Years which was purchased from Meritics, it suffered its first breakdown. Upon contacting Meritics, I was instantly put through to an engineer who was very helpful. Upon inspection of the manual, it was decided that it would be more cost effective to have a Meritics engineer visit. The Meritics staff were very communicative and kept me up to date.
-
Excellent Service

Lovely, friendly and extremely helpful. David and his team are always on call with advise and are very easy to work with and are always happy to help.
NaomiT-64
-
Service of lab equipment

Excellent service. Friendly and professional service was provided in a timely manner to the expected standard.
NathanM-270
-
Excellent

Excellent communication. Updates given well in advance. Very polite staff and engineers.
NathanM-270
-
Multisizer 4e install at pharmaceutical company

2 people helped with the install of a Multisizer 4e. They were both very professional and friendly, answering any questions that I had. The installation was quick and the attention to good documentation practice was exceptional.
Chloel-9
-
Service of Beckman Coulter instruments

Always a pleasure to welcome the Meritics service engineer, who is knowledgeable, personable and extremely competent.
NeilT-394
-
Professional

Friendly and collaborative staff, provided a very good service.
SamK-403
-
Professional, Customer-oriented, fast service

We had issues with one of the aperture tubes provided by Meritics on a Friday afternoon, while very pressed for a review deadline. Meritics handled our inquiries very fast and professionally and helped us out by shipping us their spare tube to use in the mean time free of charge, while we waited for our new tube to arrive. Communication was very pleasant.
VeerleB
-
Very helpful and prompt

I emailed for some technical help and also to get some spares and both the engineer (David) and purchasing (Sophie) were very prompt to respond and helpful. We do have a service contract but I appreciated being able to email the engineer directly.
AlexH-474
-
Excellent Service

Used Meritics to conduct particle size distribution by laser diffraction. Service as last time, is very quick, well priced and professional.
I would highly recommend
GavinR-101
-
Excellent and Friendly Service

Always a pleasure to deal with the people at Meritics Ltd. They understand the importance of our work and the fact that our research projects have deadlines that need to be met. Consequently, problems are dealt with very quickly and professionally and they are willing to go that one step further to make sure their supply chain problems don’t become our problems. Thank you to the team for their support. It is greatly appreciated.
JanetH-255