
Characterising PNIPAm Thermal Sensitive Hydrogel
Introduction to characterising PNIPAm thermal sensitive hydrogel
PNIPAm, Poly(N-isopropyl acrylamide), is a thermal sensitive polymer, which has been extensively studied since the 1990s. Due to the characteristics of its molecular structure, PNIPAm shows a phase transition corresponding to temperature variation. When the temperature exceeds its low critical solution temperature (LCST), the polymer chains gradually shrink to be a collapsed conformation because of hydrogen bond interaction and hydrophobic effect. Interestingly, either compounded or copolymerised with other materials, the thermal sensitive property of PNIPAm molecules can be effectively maintained, and the phase transition behaviour is reversible. Such a thermal sensitive characteristic is promising for many applications in pharmaceuticals and smart materials design.
In this application note, we utilized a light scattering technique to characterize the sizes and zeta potentials of a PNIPAm hydrogel with the change of temperature and studied the impact of the solution environment on its structure.
Experimental
The PNIPAm hydrogel is dispersed in water. The measurement temperature range was set to 25°C – 50°C, and measurements were performed with a temperature interval of 1°C via the function of temperature trend. A heating-cooling cycle was performed. To ensure that the measured sample is in thermal equilibrium, we set 60 seconds as the temperature equilibration time for every temperature interval.
Results and Discussion
As shown in Figure 1, during the temperature raising process, in the range of 25°C – 50°C, the particle size of PNIPAm hydrogel decreases when temperature increases, while the count rate gradually increases. At 25°C, the particle size is approximately 700 nm, while when it eventually reaches 50°C, the particle size is reduced to around 350 nm.
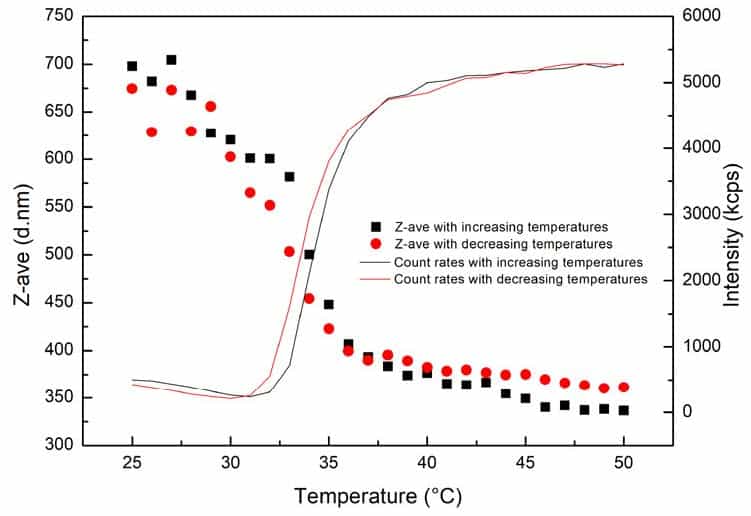
Figure 1. Particle sizes and count rates of PNIPAm hydrogel as a function of temperature.
The PNIPAm size decreases upon heating when the surrounding temperature has exceeded a phase transition temperature (which is around 32°C, as reported in most literature, but it also relies on the colloidal structure). The hydrophobic effect of PNIPAm molecules and the formation of a protein-like hydrogen bond would result in rapid changes in its conformation from the unfolded-coil state to the collapsed state. Additionally, the gradual increase of count rates results from the shrinking of PNIPAm, which leads to an increase in colloidal density and the value of (dn/dc) for suspension. And the count rates are directly proportional to the value of (dn/dc)2 .
As depicted in Figure 1, the assembled behaviour in the heating-cooling cycle is reversible. Obvious hysteresis of the phase transition can be observed in the heating and cooling cycle. It is because PNIPAm needs to absorb energy in order to form the hydrogen bonds upon heating, while the cooling process is attributed to the released energy from the breakings of hydrogen bonds.
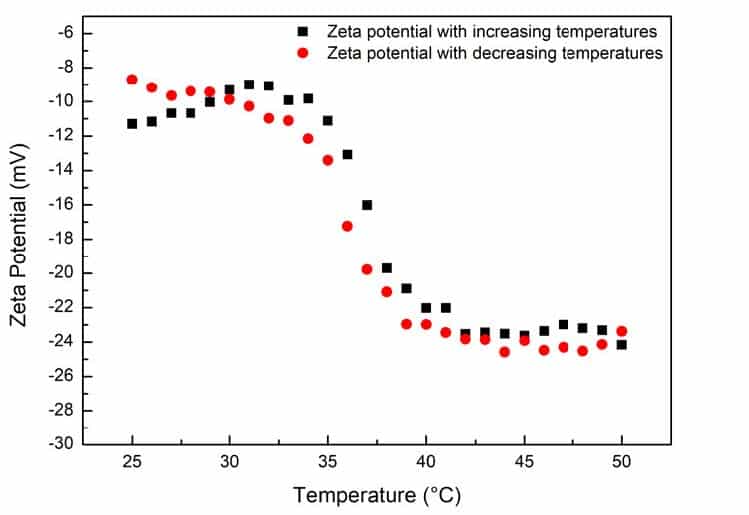
Figure 2. Zeta potentials on PNIPAm hydrogel as a function of temperature.
Figure 2 shows the zeta potentials trend of the sample under different temperatures. As illustrated, the zeta potentials of PNIPAm hydrogel are negative in the detected temperature range, which means the sample carries negative charges on the surface, and the amplitude of zeta potentials increases with the elevation of temperature. At 25°C, the zeta potential of PNIPAm is approximately -10 mV, and when the temperature reaches 50°C, the potential is increased to approximately – 24 mV. The zeta potential trend of PNIPAm hydrogel behaves the same for both the heating and cooling processes.
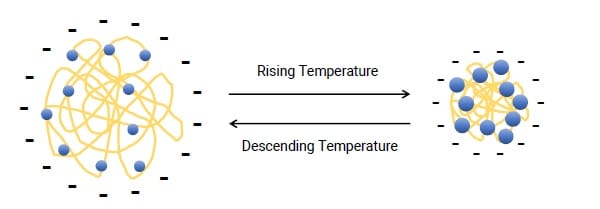
Figure 3. The schemaric diagram for the temperature dependence of PNIPAm conformation.
The temperature dependence of zeta potential of the PNIPAm sample can be illustrated in Figure 3. At relatively lower temperatures, the sample displays unfolded conformation, where the surface charge density is relatively lower; however, with the increasing temperature, the particle size decreases, leading to the increase of surface charge density and the zeta potential amplitude.
Conclusion
In this application note, a thermosensitive PNIPAm sample is characterised by automatic measurements of the particle sizes and zeta potentials under the programmed temperature change process of the BeNano. The PNIPAm measured exhibits similar behaviour with the reported results from most literature. The temperature trend measurement of the BeNano can significantly improve the measurement efficiency and provide a robust and powerful testing tool for such applications.
