
nCS1 – Exosome Application Note
Analysis of exosome concentration in blastocyst culture media by Microfluidic Resistive Pulse Sensing (MRPS) correlates with embryo implantation capacity: A pilot study
Introduction
Advances in in vitro fertilization have allowed top-rated fertility clinics to promise an approximately 70% chance of live birth from transfer of a single euploid embryo. Despite these great improvements, approximately one third of euploid embryos fail to implant.
Exosomes have recently been suggested to play roles in embryo implantation. However, because embryos are grown in a low volume of complex media (typically < 25 microliters), accurate quantification of exosomes in embryo culture has been challenging. In this early-stage pilot study, Microfluidic Resistive Pulse Sensing (MRPS), was used to predict embryo implantation by quantifying exosomes in the spent culture media of 20 human embryos.
The MPRS Technique
Microfluidic Resistive Pulse Sensing (MRPS) has recently emerged as a powerful new technique for measuring the size and concentration of extracellular vesicles. MRPS uses electrical sensing to count and size particles directly and one-by-one, without the use of any optics or mathematical conversion from diffusion behaviour to size. As a result, MRPS is independent of the material properties of the particles, and measures samples accurately no matter their polydispersity.
These fundamental strengths of the MRPS technology are particularly important for measurements of extracellular vesicles, since EV samples have low index of refraction contrast with their surrounding medium, and as biological particles are inherently polydisperse. A direct comparison of Spectradyne’s nCS1 to Nanoparticle Tracking Analysis (NTA) is shown in Figure 1 below, and shows the misleading results that can be obtained when using optical methods to quantify EVs.
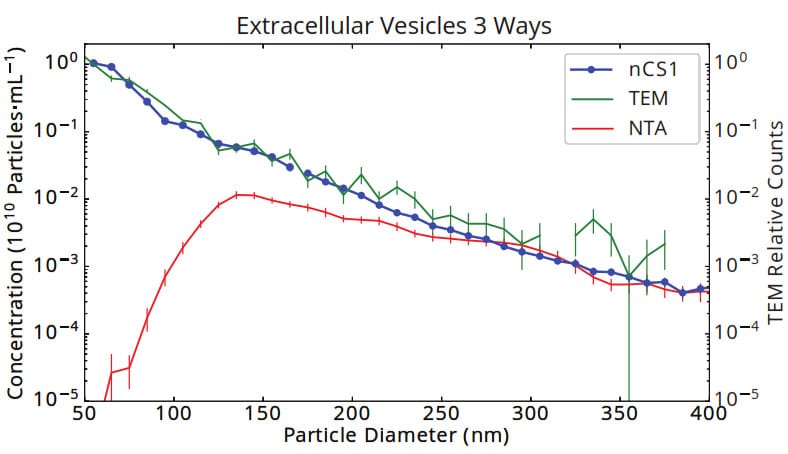
Figure 1. Size and concentration of urinary vesicles measured by Spectradyne’s nCS1, tunnelling electron microscopy (TEM) and Nanoparticle Tracking Analysis (NTA). The size distribution as measured by Spectradyne’s nCS1 agrees exactly with the gold standard, TEM, showing an approximate power-law distribution of size vs. concentration that is commonly found in EV samples. The significant loss of sensitivity of optical methods to small particles is highlighted in the NTA data, which shows a false peak at 130 nm and a discrepancy in concentration of 5 orders of magnitude at 50 nm diameter.
Practical EV Quantification
Spectradyne’s nCS1 delivers significant practical benefits that make it ideally suited for routine and quantitative EV analysis:
• Only 3 microliters sample required
• Results in minutes
• Pre-calibrated, single-use cartridges
• No cleaning required between runs
• No user-adjustable parameters
The small sample volume required for analysis directly enabled this study–only 20 microliters of culture media was available for each embryo, making analysis with any other technique impossible.
Methods
Informed consent was obtained for use of materials in this study. Spent media from blastocysts grown in single culture was collected and stored at -80°C. Spent media from 10 embryos that successfully implanted and 10 embryos that failed to implant were submitted for blinded analysis by MRPS. Samples were thawed to room temperature and 3 microliters taken from each for analysis. Total nanoparticle concentration was measured over the size range 250 nm – 2000 nm diameter and was used to predict pregnancy outcome using a threshold established from the data. As a preliminary assessment of variability in the MRPS measurements, one sample was measured in triplicate.
Results
Particle concentration showed an approximate power-law dependence on size in each sample (Figure 2). Preliminary assessment of variability in concentration measurements using one sample showed CV < 3%.
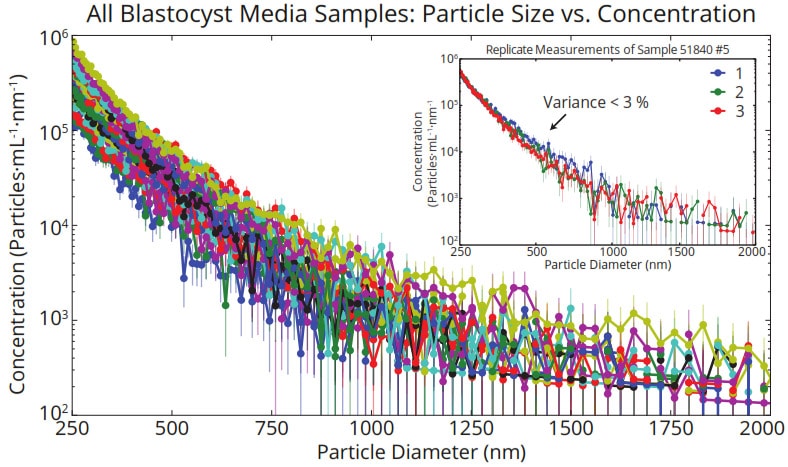
Figure 2. Size and concentration of vesicles in spent media from 20 blastocysts grown in single culture whose implantation outcomes were unknown before measurement and data analysis. Replicate measurements of a single sample (inset) yielded variance in concentration less than 3 %.
Total nanoparticle concentration across samples clustered in two groups spanning approximately 1.2×107 to 7.3×107 particles/mL, with significantly higher concentration in media from successfully-implanted embryos (Figure 3). MRPS analysis predicted pregnancy outcome with 80% sensitivity and 80% specificity.
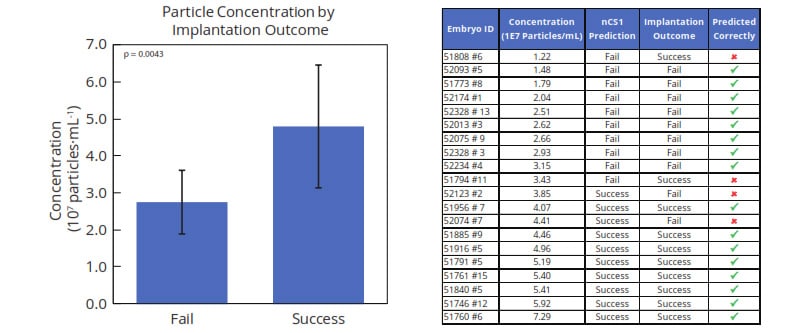
Figure 3. Total nanoparticle concentration in the range of 250-2000 nm diameter was found to be significantly higher (p < 0.005) in media of blastocysts with successful implantation outcomes. A threshold-based classification algorithm predicted pregnancy outcome with 80% sensitivity and 80% specificity.
Conclusion
In this pilot study, exosome concentrations in spent culture media measured by MRPS correlated strongly with embryo implantation potential—a tantalizing result. However, more in-depth validation is required, and consistency of measurement results must still be demonstrated more broadly. If these metrics are satisfied, MRPS could prove a valuable tool in predicting embryo implantation potential.
More information
View the Spectradyne nCS1 here.
