nCS1 – Extracellular Vesicles Application Note
Microfluidic Resistive Pulse Sensing (MRPS) validated as a rapid and practical method for evaluating Extracellular Vesicles enrichment techniques
IntroductionFor extracellular vesicles (EVs) to deliver on their promise as commercially valuable therapeutics requires improved techniques for their isolation and enrichment. However, the development of these techniques has been hindered by a lack of practical technologies for accurate EV quantification.
In this study, Microfluidic Resistive Pulse Sensing (MRPS) is validated as a rapid, practical tool for characterizing the size exclusion chromatography (SEC) method of EV purification.
The MRPS techniqueMicrofluidic Resistive Pulse Sensing (MRPS) has recently emerged as a powerful new technique for measuring the size and concentration of extracellular vesicles. MRPS uses electrical sensing to count and size particles directly and one-by-one, without the use of any optics or mathematical conversion from diffusion behaviour to size. As a result, MRPS is independent of the material properties of the particles, and measures samples accurately no matter their polydispersity.
These fundamental strengths of the MRPS technology are particularly important for measurements of extracellular vesicles, since EV samples have low index of refraction contrast with their surrounding medium, and as biological particles are inherently polydisperse. A direct comparison of Spectradyne’s nCS1 to Nanoparticle Tracking Analysis (NTA) is shown in Figure 1 below, and shows the misleading results that can be obtained when using optical methods to quantify EVs.
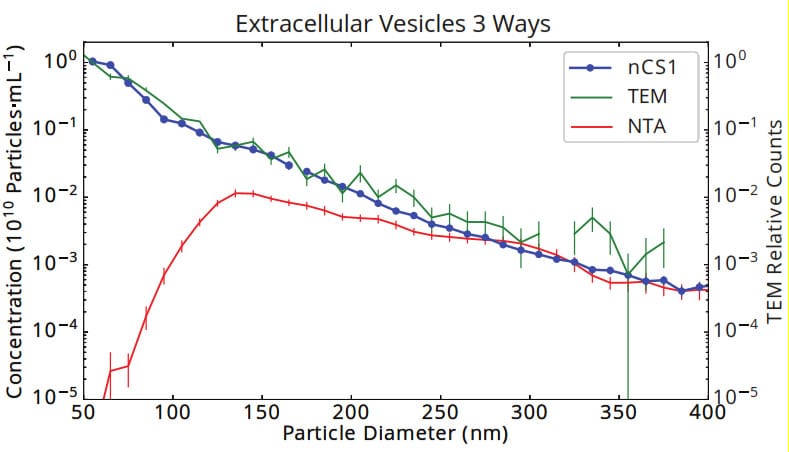
Figure 1. Size and concentration of urinary vesicles measured by Spectradyne’s nCS1, tunnelling electron microscopy (TEM) and Nanoparticle Tracking Analysis (NTA). The size distribution as measured by Spectradyne’s nCS1 agrees exactly with the gold standard, TEM, showing an approximate power-law distribution of size vs. concentration that is commonly found in EV samples. The significant loss of sensitivity of optical methods to small particles is highlighted in the NTA data, which shows a false peak at 130 nm and a discrepancy in concentration of 5 orders of magnitude at 50 nm diameter.
Practical EV QuantificationSpectradyne’s nCS1 delivers significant practical benefits that make it ideally suited for routine and quantitative EV analysis:
- Only 3 microliters sample required
- Results in minutes
- Pre-calibrated, single-use cartridges
- No cleaning required between runs
- No user-adjustable parameters
For this study in particular, the small sample volume required for analysis was a critical enabler of the measurements, since only a few tens of microliters of each sample was produced for a given set of process conditions.
MethodsDMSC25 mesenchymal stem/stromal cells were cultured to 70% confluence in growth media. Cells were then cultured for 2 days in chemically defined, vesicle-free medium. Conditioned medium (50 ml) was then concentrated by sequential ultracentrifugation and resuspended in SEC buffer and applied to a GE NAP-5 column for further purification. Fractions were collected and total EV concentration measured using MRPS on the size range of 70 nm – 400 nm. UV absorption, an orthogonal technique to MRPS, was used to quantify the total protein in each fraction. Results of each of the techniques were compared.
ResultsAs expected, MRPS measurements showed a clear peak in total particle concentration in column fractions 3-5, in which EVs are known to elute. Importantly however, particle size distributions obtained by MRPS showed that each eluted fraction contained a broad range of particle sizes spanning the full measured range of 70 nm – 400 nm, and that elution in different fractions did not significantly affect the size distribution profiles.
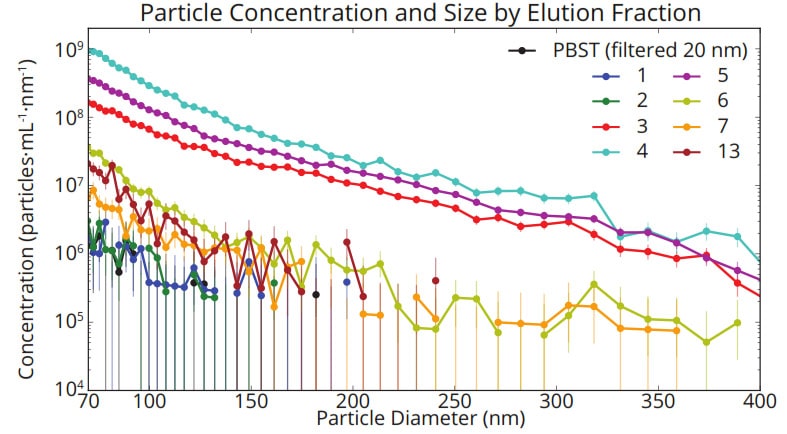
Figure 2. Concentration vs size distributions for each eluted fraction measured with the nCS1. In each fraction, a broad distribution of particle sizes is observed whose profile is independent of elution time.
Significant differences were observed between the two techniques for measurements of the non-EV fractions: a peak in total protein was detected in fractions 7 and 8, while no corresponding peak in particle concentration was observed, suggesting the protein in these fractions was not bound in the form of solid particles (Figure 3).
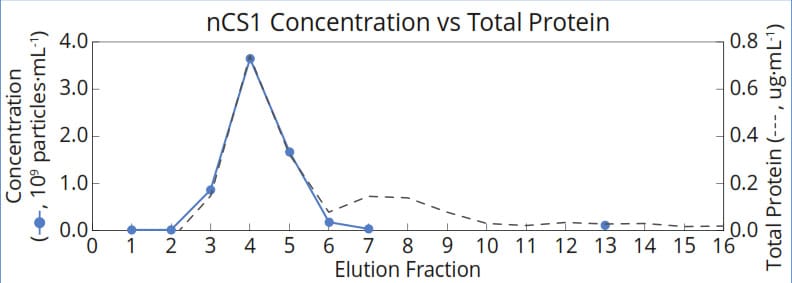
Figure 3. Measurements of nanoparticle concentration correlate strongly with total protein in fractions 1-6, in which exosomes are expected to elute. In fraction 7, nCS1 measurements indicate that protein eluting from the column is not associated with particles in this size range, but may instead be free in solution.
ConculsionMRPS was validated as a practical tool for characterising EV purification methods. In addition to demonstrating good agreement with orthogonal techniques, MRPS provided important insight about the limitations of SEC as a size-separation technique—broad particle size distributions were observed in each fraction. MRPS is therefore an important tool for accurate characterisation of EV purification methods as they are being developed.
More information
View the Spectradyne nCS1 here.
