P4SPR – Gene Regulation Application Note
P4SPR – Lac Operon / Lac Repressor(DNA-Protein binding)
Introduction
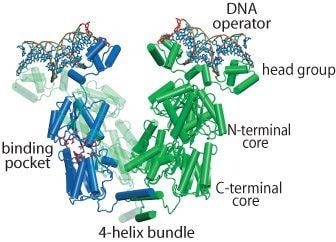
Figure 1. Lac operon binding to Lac repressor protein
The regulation of lac operon (lacO), a set of Escherichia Coli genes responsible for the lactose metabolism in the bacteria, by the lac repressor is a well-studied system in molecular biology. The lac repressor, a DNA-binding protein (lacl), is key to controlling the lac operon. The equilibrium dissociate constant (KD) of this system is estimated to be in the nM range.
This lacO / lacI system is often introduced in the context of gene regulation in biochemistry and biology class at undergraduate level. More advanced research on lacO / lacI include applications in imaging and chromatin editing.
In a broader context, SPR studies of protein-DNA complexes are important in understanding gene expression regulation pathways.
The binding strength (affinity, KD) of the lacO / lacI system was determined with the P4SPR in an academic lab at the Université de Montréal. The easy to use P4SPR and user-friendly interface allowed lab technicians to develop an SPR assay to demonstrate this biological system in undergraduate lab classes.
Experiment setup:
1. System Setup
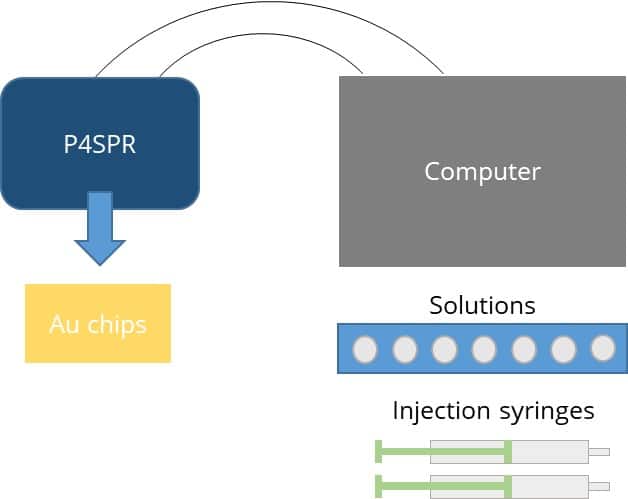

Figure 2. Benchtop P4SPR set-up
The USB-powered P4SPR can be connected to a laptop to initiate operation control and data recording using the P4SPR Control software. With its user-friendly design, the P4SPR can be set up easily. The microfluidic cell (Figure 3.) is capped over the gold sensor chip and tightly secured in the sensor chip cavity before injecting solutions in both sample and reference channels for baseline stabilization. The S-shaped sample channels has 3 parallel sensing area which can provide triplicate sample measurements along with one control measurement with the reference channel. Bubbles formed in the channels can be visually seen through the exposed clear channels for immediate removal to prevent any loss of signal.
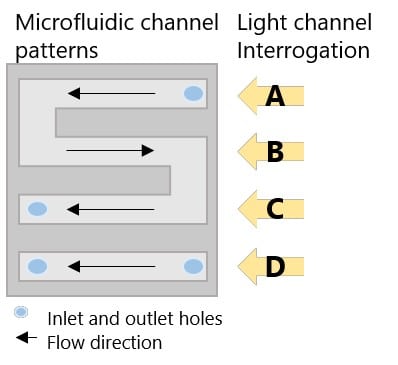
Figure 3. P4SPR microfluidic cell.
2. Experimental procedures
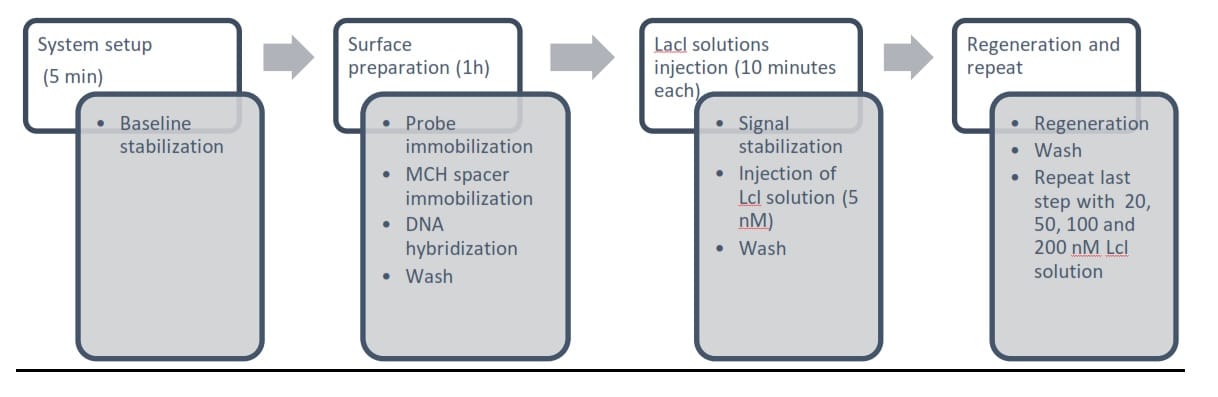
Figure 4. Experimental workflow.
After baseline stabilization has achieved, thiolated lacO DNA was injected into the sample channel. The immobilization occurred spontaneously with the strong gold-sulfur bond formation until surface saturation showed by the SPR signal plateau. The complementary lacO oligomer was then injected to allow for hybridization. The sensor surface was then washed thoroughly before the binding assay. A stock lacl protein solution was used to prepared solutions of 5, 20, 50, 100 and 200 nM. Then, each solution of increasing concentrations were sequentially injected and incubated for 10 minutes with abundant washing solution and regeneration buffer in between to remove remaining lacl proteins from previous solution.
The data from all 4 channels was recorded and then exported to Excel for further data analysis.
Results and Discussion
In this experiment, 3 parallel binding curves were generated from the samples channels. The signals were averaged and subtracted from the reference channel signal. The resulting SPR signal is plotted against time (Figure 5).
The binding shifts corresponding to different concentrations of lacl are used to establish the binding isotherm and hence determine the KD (Figure 6).
The KD was calculated to be 6.4 ± 1.2 nM which is in concordance to literature reported value in the same low nM range for the lacI-based transcription repressor proteins. It is important to note that in comparison to the DNA binding assay used in the published paper, the SPR assay did not require any DNA radiolabeling and binding interactions were monitored in real time.
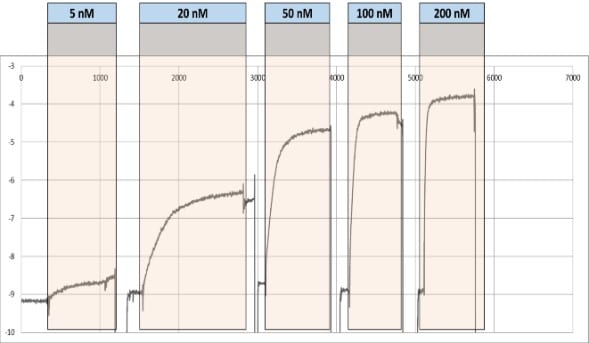
Figure 5. SPR signal with respect to different concentrations of lacO over time.

Figure 6. Superimposed binding curves of increasing lacL concentrations.
The P4SPR advantages
The unique modular, mobile and multichannel design of P4SPR makes it an accessible and flexible benchtop SPR tool for drug discovery, bio/sensing and system integration applications. It requires little to no sample preparation which enables rapid and simple system setup, while saving users cost on purification kits and reagents. Additionally, the P4SPR takes advantage of the high sensitivity and deeper penetration depth of thin film SPR for accurate and binding affinity determination.
Conclusion
The P4SPR successfully characterized the binding interaction of the lac operon DNA and its repressor protein and the result was validated with published literature. This experiment allowed undergraduate students to familiarize themselves with the technology and its application in monitoring biomolecular interactions. It is important to note that the system can also be an useful tool to characterise other biological system such as DNA-DNA, RNA as well as aptamer pairs, tailored to help scientists to better understand binding and ultimately, uncover function in a biological pathway.
Acknowledgements
We would like to thank Philipe Lampron, Shona Teijeiro and Sébastien Truche of the Université de Montréal Biochemistry department for developing the assay.
About Affinité Instruments
Established in 2015 as a spin-off of the Université de Montréal, Affinité instruments’ foundation is built deep knowledge accrued on over a decade of research in SPR. The commercialization of promising innovations is spearheaded by a diverse leadership experienced in business, science and engineering.
