Quality Control for Biopharmaceutical and Pharmaceutical Product Development
FlowCam is used for quality control and quality assurance in all stages of drug development and manufacturing. Hundreds of biopharmaceutical and pharmaceutical companies worldwide are using FlowCam to improve the quality and safety of their formulations.
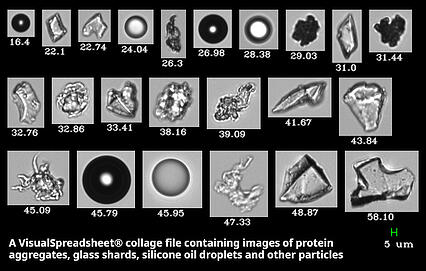
FlowCam captures high-resolution digital images of particles suspended in a fluid. Over 40 physical parameters are measured from each image, while the data is processed using our image-recognition statistical analysis software VisualSpreadsheet. FlowCam uses advanced thresholding technology to enable detailed protein analysis from 300 nm to 10+ µm.
Use real digital images to verify the size, shape, and identity of your particles. Differentiate between air bubbles, silicone oil droplets, protein aggregates, and contaminants, and determine which particles are inherent, intrinsic, and extrinsic.
In order to fully characterize the particles in your parenteral drug formulation, Flow Imaging Microscopy (FIM) is recommended by USP as a high-throughput orthogonal method to non-imaging particle analysis technology like light obscuration and dynamic light scattering.
- Analyze particles 300 nm to 10+ µm
- Perform statistical calculations
- Generate size distribution curves
- Visually verify particle identity with high-resolution images
- Measure 40+ physical parameters per particle
- Meet USP 787/788 Guidelines
- Perform 21 CFR Compliance
- Require only 100 µl minimum sample volume
“FlowCam paid for itself in just one incident. Suddenly our [parenteral drug] began to show visible particles. So, we FlowCammed a sample from each step of the manufacturing process and saw that one of our filling pumps was shedding metal shards, catalyzing aggregation. We were able to fix the pump and continue with manufacturing. In just that one instance, FlowCam saved us time, money, and enabled manufacturing to continue that same day.”
— Biopharmaceutical FlowCammer, USA, 2018
