
Revolution – Pharmaceutical Application Note
The Effect of Lubricants on Powder Flowability for Pharmaceutical Application
Abstract
Pharmaceutical tablets are manufactured through a series of batch steps finishing with com- pression into a form using a tablet press. Lubricants are added to the powder mixture prior to the tabletting step to ensure that the tablet is ejected properly from the press. The addition of lubricants also affects tablet properties and can affect the behaviour of the powder mixture. The objective of this research was to investigate the effect of lubricants on powder flowability as flowability into the tablet press is critical. Four lubricants (magnesium stearate, magnesium silicate, stearic acid, and calcium stearate) were mixed, in varying amounts, with spray-dried lactose. In addition, magnesium stearate was also mixed with placebo granules from a high-shear granulator. Measurements based on avalanche behaviour indicated flowability potential and dynamic density and were more sensitive to changes in the mixture and provided a more accurate and reproducible indication of flowability than traditional static measurements. Of the tested lubricants, magnesium stearate provided the best increase in flowability even in the low amounts commonly added in formulations.
Introduction
Lubricants are commonly added to help in the tabletting of many formulations. Following compression, a tablet must be ejected out of the tablet press die. Lubricants reduce the friction between the tablet and the die metal surface, which reduces the ejection force and helps to ensure that the tablet is cleanly ejected and without cracking or breakage.
There are two ways in which lubricants reduce friction: a liquid lubricant can form a thin, continuous fluid layer be- tween the tablet and the metal die surface or lubricant particles can form a boundary layer on the formulation particles or metal die surfaces. Boundary lubricants are more commonly used over fluid lubricants (1). There are six types of commonly used boundary lubricants for pharmaceutical tableting: (a) metallic salts of fatty acids; (b) fatty acids, hydrocarbons, and fatty alcohols; (c) fatty acid esters; (d) alkyl sulphates; (e) polymers; and (f) inorganic materials (1).
Magnesium stearate, a metallic salt boundary lubricant, is probably the most commonly used lubricant for pharmaceutical tableting; it is relatively inexpensive, provides high lubrication, has a high melting point, and is chemically stable (2). Calcium stearate is another metallic salt boundary lubricant that can replace magnesium stearate in a formulation (1). Metallic salt lubricants are typically added to formulations in the range of 0.25 to 1.0 wt.% (1). Stearic acid is the most commonly used fatty acid boundary lubricant. In general, fatty acids are more effective die lubricants than the corresponding alcohols, and the alcohols are better than the corresponding hydrocarbons (3). Stearic acid is typically added at levels of approximately 2.5 wt.% (1). Hydrous magnesium silicate or talc is an inorganic boundary lubricant. It is useful when other lubricants cannot be used due to chemical instabilities and is typically added in the range of 1.0 to 10.0 wt.% when being used as a tablet glidant and/or lubricant (1, 4–6).
Lubricants are necessary for tabletting, but they can cause undesirable changes in the properties of the tablet. Lubricant type, concentration, method of lubrication, and the manner of incorporating the lubricant all affect tablet compression (7). It is generally accepted that magnesium stearate has more negative effects on the hardness of the tablets with more deformable materials than brittle ones (1). When microcrystalline cellulose, an example of a plastic material, was mixed with magnesium stearate, the tablet strength was weakened significantly as the amount of added lubricant in- creased. Similar results were obtained with other lubricants (8). The adverse impact of magnesium stearate on other commonly used plastically deformable excipients, such as lactose and starch, was also observed (9). Since many lubricants are hydrophobic, tablet disintegration and dissolution rates are often reduced by the addition of lubricant (3). Multiple studies (10–12) have concluded that the negative effects are due to a combination of the large surface area and hydrophobicity of the lubricant.
Many methods have been developed to measure the flow properties of powder. These methods commonly include the static angle of repose, Carr’s compressibility index, Hausner ratio, and shear cell testing. Avalanche testing is an emerging powder flowability measurement method. The avalanche testing equipment consists of a rotating drum and measurement system. As the drum rotates, the powder sample is carried up the side of the drum until the weight of the powder causes it to collapse or avalanche. Properties of this powder avalanche are then measured and analysed to indicate powder flowability. The measurement methods and analysis vary with the manufacturer: a photocell array for the Aero-Flow Analyzer, an advanced camera and imaging system for the Revolution Analyser (13), a load cell to measure the change in moment of inertia for the Gravitation Displacement Rheometer (14) and positron emission particle tracking for equipment developed by Lim et al. (15).
Powder flow is critical during tableting as it must flow easily and uniformly into the tablet dies to ensure tablet weight uniformity and production of tablets with consistent and reproducible properties (16–18). There are many factors that influence powder flow. Examples of physical properties that influence powder flow include particle size and size distribution, density, surface morphology, and particle shape. In general, smaller particles with low densities and an irregular surface and shape exhibit decreased flow compared to large, smooth, and spherical particles with high densities (19). Particle shape and surface morphology affect particle–particle con- tact and therefore can increase friction if contact area is increased which then reduces flowability. As boundary layer lubricants form a film around particles, these lubricants can affect friction through modified particle–particle contact, thereby also affecting powder flow.
Although the primary role of lubricants is to improve tabletting, and of glidants is to improve flowability into the tablet press, some lubricants can also improve flow. The effect of lubricants on flowability has been examined by several researchers. Using the Hausner ratio and shear cell measurements, Liu et al. (20) found that the addition of 0.5 wt.% of magnesium stearate improved the flowability of cohesive ibuprofen. Faqih et al. (14) examined the avalanche behaviour of commonly used pharmaceutical powders mixed with varying amounts of magnesium stearate. The effect of magnesium stearate on the flowability varied with the powder: there was almost no effect on the already free-flowing Fast Flo lactose, but a significant effect on the very cohesive regular lactose. Pingali et al. (21) examined the flow behaviour of acetaminophen, didesmethylesibutramine tartrate, and levalbuterol tartrate after mixing with lubricants colloidal silica, talc, and magnesium stearate. A flow index obtained from avalanche behaviour showed that the flowability increased with the addition of the lubricants.
As magnesium stearate is the most commonly used boundary layer lubricant, it has been the most extensively studied. It has been hypothesized that the film formed by magnesium stearate around other excipient particles could be: (a) as a monomolecular film of magnesium stearate particles bound to the other excipients by their a polar heads, (b) as a monoparticulate film of magnesium stearate particles covering the surface of the other excipients, and (c) as layers of magnesium stearate particles first filling any cavities of the other excipients before forming a continuous layer (2, 22). The third hypothesised mechanism of first filling any cavities is the most popular. With this mechanism, magnesium stearate would improve flow by minimizing any surface irregularities of the excipients, which reduces contact points between excipients, decreasing friction, and cohesive forces.
Although lubricants are necessary and added to help tabletting, their impact upon tablet properties and manufacturing cannot be ignored. Many studies have examined the effect of lubricants on tablet properties. Only a few have examined the effect on flowability. As flowability is critical to many operations including tabletting, the objective of this research was to examine the impact of lubricants on the flowability of pharmaceuticals. Compared with other studies, this research extensively examined the effect of many lubricants at different amounts on flowability measured using a variety of techniques. Initially, spray-dried lactose was used as the test particle. Further trials were then conducted with placebo granules from a high-shear granulator.
MATERIALS AND METHODS
Test Particles
Spray-dried lactose (FlowLac 100, Meggle) was used as the initial test particle. As dried granules are commonly mixed with lubricants just before tabletting, the spray-dried lactose was then replaced with placebo granules. The granules were made in a high-shear granulator from 50% (w/w) (dry basis) lactose monohydrate (Merck, dpsm of 113 μm), 45% (w/w) microcrystalline cellulose (Alfa Aesar, dpsm of 77 μm), 4% (w/w) hydroxypropyl methylcellulose (Alfa Aesar, dpsm of 48 μm), and 1% (w/w) croscarmellose sodium (Alfa Aesar, dpsm of 139 μm) with water as a liquid binder added to a moisture content of 30 wt.%. Granules were then oven dried at 24°C and a relative humidity of 2 to 3% for more than 24 h to ensure a granule moisture content of less than 2 wt.%. Dried granules were sieved to include only those between 150 and 600 μm.
Lubricants
Four lubricants were blended with the spray-dried lactose up to 5 wt.%. Only small amounts of lubricants are added to formulations. Trials included lubricant additions beyond commonly used amounts to provide easier identification of any trends and differences between lubricants. Magnesium stearate was blended with the granules up to 5 wt.%.
Mixing Process
The powders were blended using a Patterson Kelly V blender with a stainless steel V shell for 15 min at a rotation speed of 25 rpm.
Mixture Analysis
Particle Size
Particle size distributions of the spray-dried lactose and lubricants were measured using a Malvern Mastersizer 2000.
Shape and Morphology
Scanning electron microscope (SEM) images of the spray-dried lactose, granules, lubricants, and samples of the mixtures were taken using a Hitachi S-4500 field emission SEM. The powders were mounted on a plate and coated with gold before examination. The images allowed the shape and morphology of the powders to be examined and allowed visualization of the interaction of the lubricants with the spray-dried lactose and granules.
FlowabilityDensity
The bulk and tapped densities of the mixtures were measured using 100-ml samples. For the bulk density measurements, the powder flowed down a vibrating chute into a 100- ml cylinder and the mass of the powder sample within the cylinder was then measured as follows:
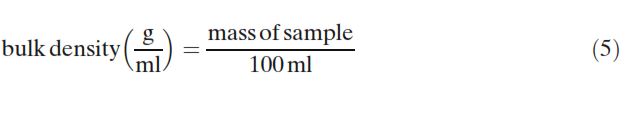
The sample within the cylinder was then vibrated/tapped until the resulting volume no longer changed and was then measured to determine the tapped density:
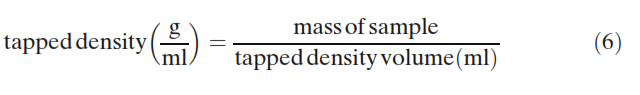
Duplicate density measurements were performed. The bulk and tapped density measurements then allowed the Hausner ratio and Carr index to be calculated (23):
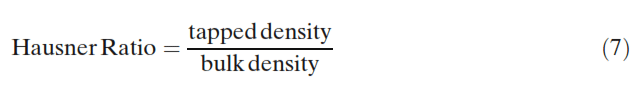
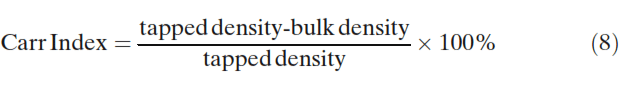
Static Angle of Repose
Static angle of repose measurements were performed using a Powder Research Ltd. angle of repose device. Samples of about 60 ml flowed down a vibrating chute and through a funnel to form a pile below on a calibrated level platform to allow the angle of repose to be determined. Samples were measured in triplicate.
Avalanche Behaviour
Various indicators of flowability were investigated using a Mercury Scientific Revolution Powder Analyser. A sample size of 118 cm3 was loaded into a drum with a diameter of 11 and width of 3.5 cm. This drum was rotated at 0.3 rpm until 128 avalanches had occurred; an avalanche was defined as being a rearrangement of at least 0.65 vol.% of the sample in the drum. Optical measurements with a resolution of 648× 488 at 60 frames per second monitored the powder surface as the sample was rotated and software calculated the flowability indicators. Samples were measured in triplicate.
The two main indicators of flowability analysed were avalanche time and dynamic density. Avalanche time is determined as the average amount of time between successive avalanches during drum rotation. The Mercury Scientific Revolution Powder Analyzer also measured the dynamic density of powders through the known sample weight and the measured volume of the powder as it tumbled within the drum.
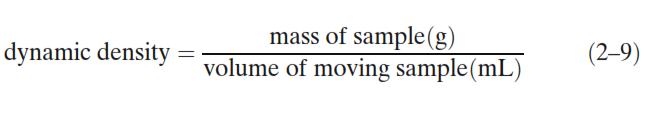
RESULTS
Size and Size Distribution
The size distributions of the spray-dried lactose and the lubricants are given in Figs. 1 and 2, respectively. The magnesium silicate, magnesium stearate, and calcium stearate, were all smaller than the spray-dried lactose. Stearic acid was the largest tested lubricant: some of its larger particles were similar in diameter to the spray-dried lactose.
Visual Observations
Figure 3 shows scanning electron images of the spray dried lactose and the lubricants. The spray-dried lactose particles were approximately spherical with a slightly textured surface and small surface cavities. The magnesium stearate, magnesium silicate, and calcium stearate particles were all irregular flakes with a high surface area to volume ratio. In contrast, the stearic acid particles were almost spherical with a smooth surface morphology.
Figures 4, 5, 6, and 7 shows scanning electron images of samples of the mixtures of the spray-dried lactose with the lubricants; the magnesium stearate flakes appeared to first fill any cavities on the surface of the lactose particles. A continuous layer of magnesium stearate was not formed. Instead, at high concentrations, the magnesium stearate remained unattached to the lactose particles and began to form self agglomerates. Calcium stearate flakes attached to the surface of the lactose particles and at higher concentrations continued to layer around the lactose. Magnesium silicate first filled any surface cavities, but then continued to layer around the spray dried lactose particles and also formed self-agglomerates. Regardless of concentration, most stearic acid particles remained unattached to the lactose.
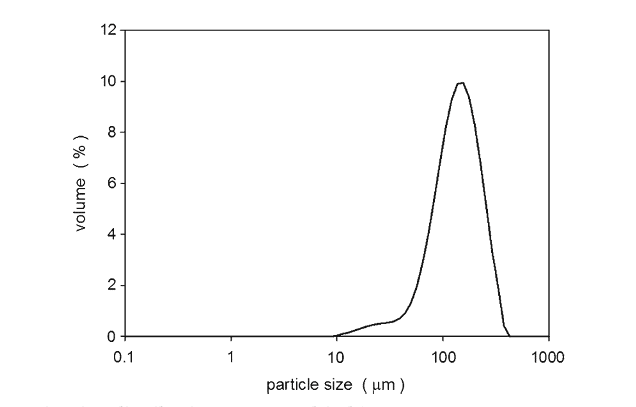
Fig. 1. Size distribution of spray-dried lactose
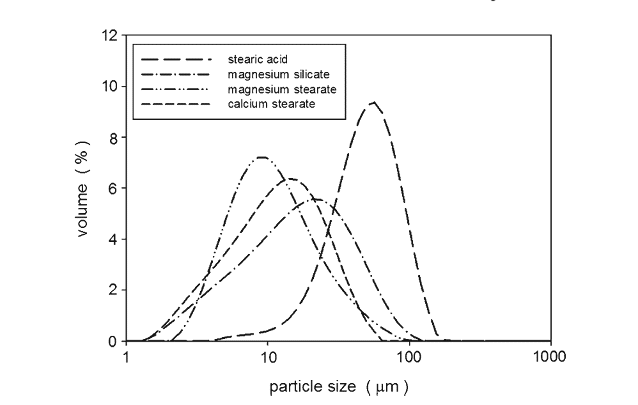
Fig. 2. Size distributions of lubricants magnesium stearate, magnesium silicate, calcium stearate, and stearic acid
Flowability
Static Angle of Repose
Figure 8 shows the measured static angle of repose for the various lubricant mixtures. Spray-dried lactose showed good flowability with a static angle of repose of about 32°. The addition of the lubricants decreased the static angle of repose indicating an improvement in flowability. The flowability, however, did not continuously improve as more lubricant was added to the mixtures. For magnesium stearate, magnesium silicate, and stearic acid, the flowability improved up to 1–2 wt.% of lubricant but then, further lubricant additions up to 5 wt.% had no significant impact on the flow. Calcium stearate mixtures showed a different profile for the static angle of repose: the angle decreased to 27° at 0.5 wt.% calcium stearate but then increased to 29.5° at 3 wt.% with further additions having no significant effect on the flow.


Fig. 3. SEM images of raw components (a magnesium stearate, b magnesium silicate, c calcium stearate, d stearic acid, e spray-dried lactose)


Fig. 4. SEM images magnesium stearate and spray-dried lactose mixtures (a 0.5, b 1.0, c 1.5, d 2.0, e 3.0, f 5.0 wt.%)
Density Measurements
The Hausner ratio and Carr index use the tapped and bulk densities to indicate flowability. Some trends with increasing lubricant addition were observed, but the Hausner ratio and Carr index did not provide enough resolution to form conclusions about changes in flowability.
Avalanche Time
The avalanche time is a flowability indicator measured by the Mercury Scientific Revolution Powder Analyzer. Figure 9 shows that the avalanche time decreased with the introduction of the lubricants. The avalanche time for the mixture with magnesium stearate and magnesium silicate decreased to an avalanche time near 3 s by 2–3 wt.% of the lubricant. Calcium stearate decreased the avalanche time to a low value of 2.5 s at a concentration of only 1 wt.%, but the addition of more lubricant increased the avalanche time. The addition of stearic acid to the spray-dried lactose initially slightly decreased the avalanche time. Larger amounts of stearic acid, however, showed no significant changes in the avalanche time.
Dynamic Density
The dynamic density shown in Fig. 10 was also measured by the Mercury Scientific Revolution Powder Analyzer. The addition of magnesium stearate increased the dynamic density from about 0.63 to a maximum of 0.69 g/ml near 2 wt.% addition of magnesium stearate. Further addition of magnesium stearate decreased the dynamic density slightly. Calcium stearate provided a large increase to a density near 0.83 g/ml. There were, however, no significant changes in the dynamic density with the additions of magnesium silicate or stearic acid.
Magnesium Stearate Interaction with Granules
The scanning electron micrographs in Fig. 11 show the interaction of the magnesium stearate with the granules with the magnesium stearate preferentially filling the large cavities of the granules. Both the static angle of repose and the avalanche time (Fig. 12) indicated that the magnesium stearate addition improved the flowability of the granules.


Fig. 5. SEM images calcium stearate and spray-dried lactose mixtures (a 0.5 wt, b 2.0 wt, c 4.0 wt.%)


Fig. 6. SEM images magnesium silicate and spray-dried lactose mixtures (a 0.5 wt, b 2.0, c 3.0 wt.%)
DISCUSSION
The scanning electron micrographs in Fig. 4 showed that the magnesium stearate flakes first filled the surface cavities of the spray-dried lactose. At levels above 1.5–2 wt.%, the magnesium stearate no longer attached to the spray-dried lactose. Instead, the flakes began to form self-agglomerates. This critical point of 1.5–2 wt.% was observed as a transition point in the flowability measurements. The static angle of repose decreased from 32 to 27° by 1.5 wt.% (Fig. 8a). According to the flowability relationships summarized by Nagel and Peck (24), this indicated that the addition of the magnesium stearate improved the flow from good to excellent. The avalanche time also decreased from 4.4 to 3.1 s by 2 wt.% magnesium stearate addition indicating an increase in the flowability (Fig. 9a). The magnesium stearate filled the surface cavities of the spray dried lactose. This selective adhesion to the surface improved the flowability of the powder by improving the surface of the lactose particles: the particles became more spherical with a smoother surface morphology, which decreased the frictional forces by reducing contact points, thus improving flow (2, 22).
The selective adhesion of magnesium stearate to the surface cavities is also supported by the dynamic density measurements (Fig. 10a). The dynamic density increased from 0.63 to 0.69 g/ml at 2 wt.%, but further additions of magnesium stearate reduced the density to 0.67 g/ml. Filling the surface cavities of the lactose with the magnesium stearate increased the weight of each lactose particle without adding any bulk volume. The agglomerates of magnesium stearate that began to form after 2 wt.% then began to change the bulk volume of the powder mixture, which decreased the dynamic density.
Although calcium stearate is also a metallic salt boundary lubricant and similar to magnesium stearate, its behaviour and effects when added to the spray-dried lactose were different.

Fig. 7. SEM images of stearic acid and spray-dried lactose mixtures (a 1.0, b 3.0 wt.%)
The scanning electron micrographs (Fig. 5) indicated that the calcium stearate did not preferentially fill the surface cavities of the lactose particles after approximately 1 wt.%. At higher levels, the calcium stearate continued to attach to the surface of the spray-dried lactose particles without the formation of a significant amount of self-agglomerates. As the addition of calcium stearate continued, the surface morphology of the lactose particles began to change: the adhered calcium stearate formed an irregular surface. This change in the surface irregularity increased the contact points, which increased frictional forces. The larger frictional forces decreased flowability and this was shown in the flowability measurements: the static angle of repose decreased from 32 to 27° by 0.5 wt.% addition of calcium stearate, but further additions increased the angle to 30° (Fig. 8b) and the avalanche time decreased to 2.6 s by 1 wt.%, but then increased to beyond 3.8 s with further additions (Fig. 9b).
The layering of calcium stearate on the spray-dried lactose particles was reflected in the increased dynamic density (Fig. 10b). The dynamic density increased from 0.63 to 0.85 g/ ml with the addition of calcium stearate. The adhered calcium stearate increased the weight of the lactose particles without significantly increasing the volume as the small flakes only formed a thin layer on the lactose surface.
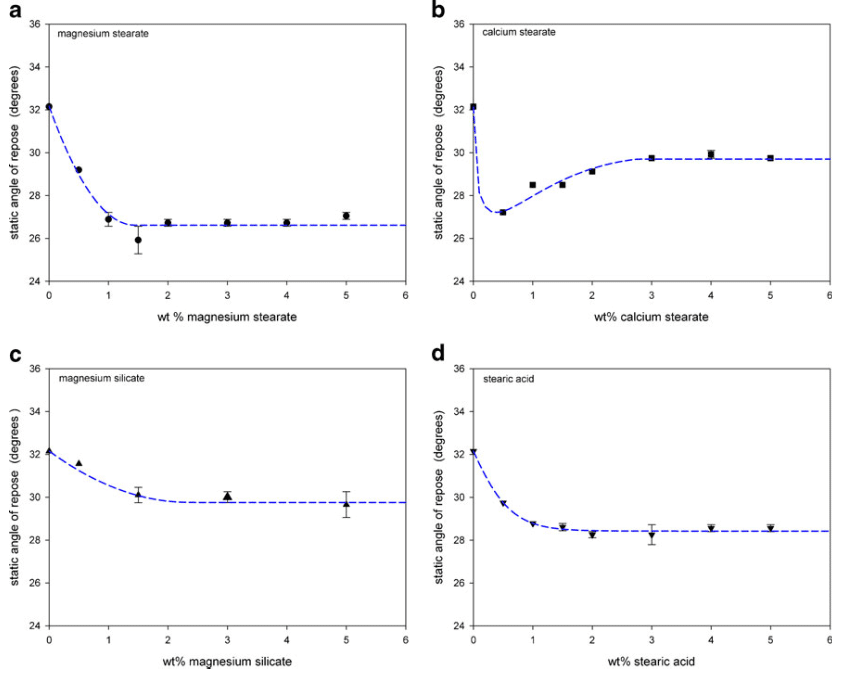
Fig. 8. Static angles of repose for the lubricant and spray-dried lactose mixtures
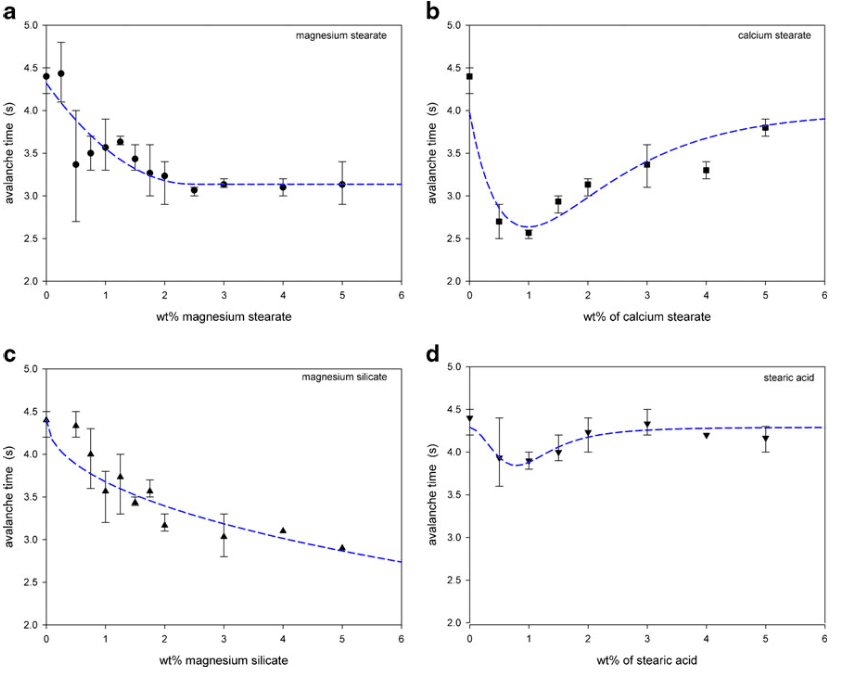
Fig. 9. Avalanche times for the lubricant and spray-dried lactose mixtures
Magnesium silicate flakes appeared to first partially fill in the surface cavities of the spray-dried lactose (Fig. 6). However, above 2 wt.%, magnesium silicate both continued to adhere to the surface of the lactose and also form self-agglomerates. The initial preferential filling of the surface cavities improved flowability by increasing the sphericity of the lactose and smoothing the surface. This was reflected in the static angle of repose which decreased slightly from 32 to 30° (Fig. 8c) and in the decrease in avalanche times (Fig. 9c). Improvements in the flowability beyond 2 wt.% were only detected through the avalanche times which continued to decrease to just below 3 s. Coating of the lactose surface would form an irregular surface increasing contact points and friction thereby decreasing flowability. However, the relatively large self-agglomerates of magnesium silicate should exhibit reasonable flowability potential. The effect on flowability by additions of magnesium silicate beyond 2 wt.% was therefore complex. Changes in the dynamic density (Fig. 10c) were difficult to detect with the varying interactions between the magnesium silicate and the spray-dried lactose.
Given its large size and spherical shape (Figs. 2 and 3), stearic acid did not fill in any surface cavities of the spray-dried lactose and it did not adhere to the surface of the lactose. As shown by the static angle of repose (Fig. 8d) and the avalanche time (Fig. 9d), stearic acid slightly improved the flowability for additions less than about 1 wt.%. The smooth and almost spherical stearic acid particles acted as spacers between the lactose particles. This reduced cohesive forces between the lactose particles and also reduced frictional forces as there were fewer stearic acid–lactose contact points than lactose–lactose contact points.
The interactions between the lubricant and the spray-dried lactose varied with the lubricant and the amount of added lubricant. With the smallest median size, the magnesium stearate easily filled the cavities on the surface of the lactose particles. At this small size, van der Waals forces are important (4, 25) and promoted the self-agglomeration of the magnesium stearate flakes which did not immediately adhere to the lactose.
Studies have been conducted showing that mixing time affects lubricating properties of magnesium stearate (26, 27). During the initial stages of mixing, magnesium stearate adheres to the other particles. As mixing continues, some magnesium stearate can detach by delamination or de-aggregation. Therefore, over time, the magnesium stearate continues to be distributed throughout the powder which negatively affects tablet properties through further increases in disintegration times and decreases in mechanical strength. Perrault et al.(2) investigated magnesium stearate mixing and expressed concerns that, in many cases, magnesium stearate may not be homogeneously mixed given the low amount usually added to a formulation combined with its cohesiveness. The variability in avalanche times at low-lubricant additions, particularly for magnesium stearate, was shown in Fig. 9. The avalanche measurements are dynamic and were conducted in triplicate with each measurement, an average of 128 avalanches over approximately four revolutions of the sample drum. Small changes in the magnesium stearate distribution in the sample were therefore detected resulting in the high variability of the avalanche times.
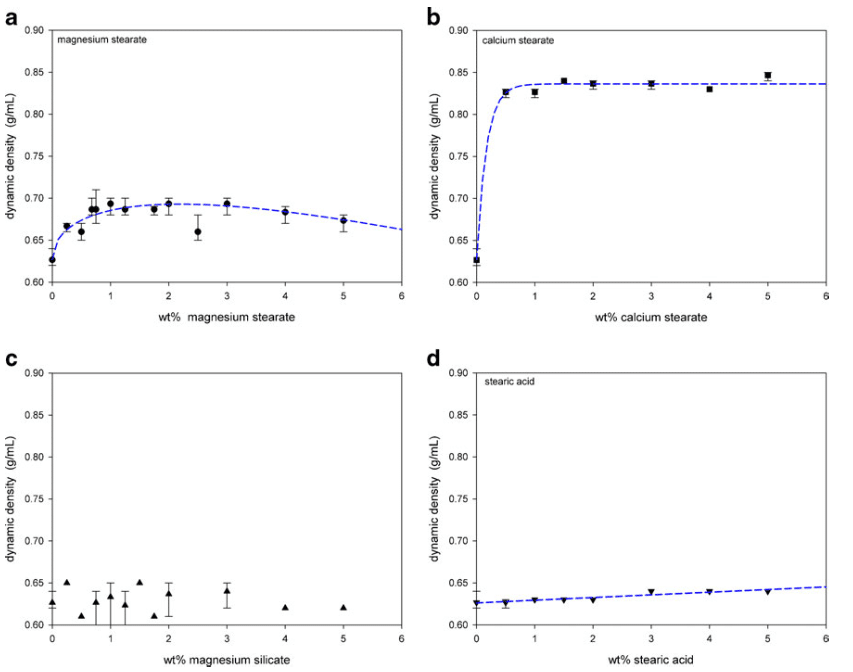
Fig. 10. Dynamic densities for the lubricant and spray-dried lactose mixtures

Fig. 11. SEM images of magnesium stearate and granule mixtures (a 1.0, b 2.0, c 3.0, d 4.0 wt.%)
Magnesium stearate interactions were similar for the granules and the spray-dried lactose, preferentially first filling cavities of both particles. Filling the large cavities of the granules smoothed the surface and created more spherical particles which decreased the frictional forces leading to improved flowability (Fig. 11). As the granules were very irregular with large cavities, the increase in flowability from the magnesium stearate interaction was significant (Fig. 12). In addition, compared to the spray-dried lactose, more magnesium stearate filled the granule cavities before beginning to form self-agglomerates at about 4 wt.%; at which point, further increases in the flowability were only minimal.
For magnesium stearate added to formulated granules, the optimal amount is usually a balance of lubrication requirements for ejection from the tablet press and negative impacts upon tablet properties such as strength and dissolution. The flowability should be measured at this point to determine improvements in flowability from the magnesium stearate.
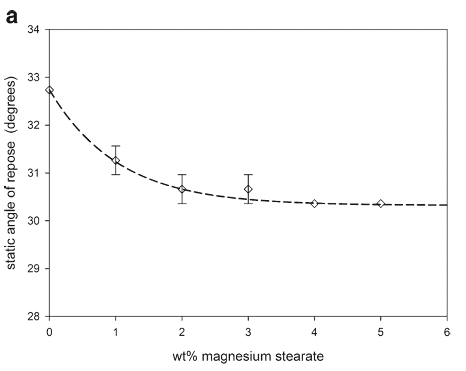
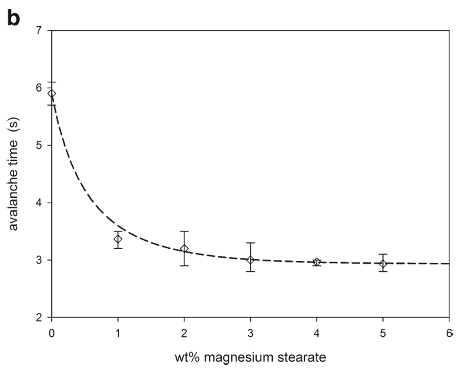
Fig. 12. Static angles of repose and avalanche times for magnesium stearate and granule mixtures
This improvement, in turn, could reduce the required amount of other excipients such as glidants. Therefore, determining the impact of magnesium stearate on flowability could promote changes in the formulation leading to a composition with fewer excipients while maintaining a high-quality tablet.
CONCLUSIONS
Lubricants were mixed with spray-dried lactose and placebo granules in varying amounts to determine the effect of lubricants on flowability. Differences in interactions between the lubricants and the particles affected flowability. Both magnesium stearate and magnesium silicate initially preferentially filled the surface cavities on the spray-dried lactose improving flowability by creating more spherical and smoother particles with less friction contact points. Calcium stearate did not seem to preferentially fill surface voids, and thus showed a different flowability effect; after an initial improvement in flow, excess calcium stearate decreased overall flow, since the excess lubricant increased surface irregularities on the spray-dried lactose. Stearic acid showed little effect on flowability. Of the tested lubricants, magnesium stearate provided the most improvement in flowability even at the low amounts that would be used in formulations. Magnesium stearate also preferentially filled surface cavities of the granules and significantly improved their flowability. As it is critical for tablet formation, the conclusions of this study highlight the importance of considering not only tablet performance and tablet final properties, but also flowability in lubricant selection.
ACKNOWLEDGMENTS
The authors would like to acknowledge the Natural Sciences and Engineering Research Council of Canada and the Ontario Graduate Scholarship for their financial support. The Western University Graduate Thesis Research Award Fund is also acknowledged for financial contributions.
REFERENCES
1. Wang J, Wen H, Desai D. Lubrication in tablet formulations. Eur J Pharm Biopharm. 2010;75(1):1–15.
2. Perrault M, Bertrand F, Chaouki J. An investigation of magnesium stearate mixing in a V-blender through gamma-ray detection. Powder Techn. 2010;200(3):234–45.
3. Alderborn G. Tablet and compaction. In: Aulton ME, editor. Pharmaceutics: the science of dosage form design. 2nd ed. London: Churchill Livingstone; 2002. p. 397–448.
4. Dawoodbhai S, Rhodes CT. Pharmaceutical and cosmetic uses of talc. Drug Dev Ind Pharm. 1990;16(16):2409–29.
5. Dawoodbhai TS, Suryanarayan E, Woodruff C, Rhodes C. Optimization of tablet formulations containing. Drug Dev Ind Pharm. 1991;17(10):1343–71.
6. Wang DP, Yang MC, Wong CY. Formulation development of oral controlled-release pellets of diclofenac sodium. Drug Dev Ind Pharm. 1997;23(10):1013–7.
7. Asker A, Saied K, Abdel-Khalek M. Investigation of some materials as dry binders for direct compression in tablet manufacture. Part 5: effects of lubricants and flow conditions. Die Pharmazie. 1975;30(6):378.
8. Jarosz PJ, Parrott EL. Effect of lubricants on tensile strengths of tablets. Drug Dev Ind Pharm. 1984;10(2):259–73.
9. Mitrevej A, Sinchaipanid N, Faroongsarng D. Spray-dried rice starch: comparative evaluation of direct compression fillers. Drug Dev Ind Pharm. 1996;22(7):587–94.
10. Johansson M. Investigations of the mixing time dependence of the lubricating properties of granular and powdered magnesium stearate. Acta Pharm Suec. 1985;22(6):343.
11. Johansson ME. Influence of the granulation technique and starting material properties on the lubricating effect of granular magnesium stearate. J Pharm Pharmacol. 1985;37(10):681–5.
12. Johansson M, Astra Laekemedel A, Soedertaelje S. The effect of scaling-up of the mixing process on the lubricating effect of powdered and granular magnesium stearate. Acta Pharm Tech. 1986;32(1):39–42.
13. Nalluri VR, Kuentz M. Flowability characterisation of drug-excipient blends using a novel powder avalanching method. Eur J Pharm Biopharm. 2010;74(2):388–96.
14. Faqih AMN, Mehrotra A, Hammond SV, Muzzio FJ. Effect of moisture and magnesium stearate concentration on flow properties of cohesive granular materials. Int J Pharm. 2007;336(2):338– 45.
15. Lim SY, Davidson J, Forster R, Parker D, Scott D, Seville J. Avalanching of granular material in a horizontal slowly rotating cylinder: PEPT studies. Powder Tech. 2003;138(1):25–30.
16. Fassihi A, Kanfer I. Effect of compressibility and powder flow properties on tablet weight variation. Drug Dev Ind Pharm. 1986;12(11–13):1947–66.
17. Tan S, Newton J. Powder flowability as an indication of capsule filling performance. Int J Pharm. 1990;61(1–2):145–55.
18. Sandler N, Wilson D. Prediction of granule packing and flow behavior based on particle size and shape analysis. J Pharm Sci. 2010;99(2):958–68.
19. Amidon GE. Physical and mechanical property characterization of powders. In: Brittain HG, editor. Physical characterization of pharmaceutical solids. Zug: Informa Healthcare; 1995. p. 281–319.
20. Liu LX, Marziano I, Bentham A, Litster JD, White E, Howes T. Effect of particle properties on the flowability of ibuprofen powders. Int J Pharm. 2008;362(1):109–17.
21. Pingali KC, Saranteas K, Foroughi R, Muzzio FJ. Practical methods for improving flow properties of active pharmaceutical ingredients. Drug Dev Ind Pharm. 2009;35(12):1460–9.
22. Roblot-Treupel L, Puisieux F. Distribution of magnesium stearate on the surface of lubricated particles. Int J Pharm. 1986;31(1):131–6.
23. Carr RL. Evaluating flow properties of solids. Chem Eng. 1965;72:163–8.
24. Nagel KM, Peck GE. Investigating the effects of excipients on the powder flow characteristics of theophylline anhydrous powder formulations. Drug Dev Ind Pharm. 2003;29(3):277–87.
25. Li Q, Rudolph V, Weigl B, Earl A. Interparticle van der Waals force in powder flowability and compactibility. Int J Pharm. 2004;280(1–2):77–93.
26. Kikuta JI, Kitamori N. Effect of mixing time on the lubricating properties of magnesium stearate and the final characteristics of the compressed tablets. Drug Dev Ind Pharm. 1994;20(3):343–55.
27. Bossert J, Stains A. Effect of mixing on the lubrication of crystalline lactose by magnesium stearate. Drug Dev Ind Pharm. 1980;6(6):573–89.
